 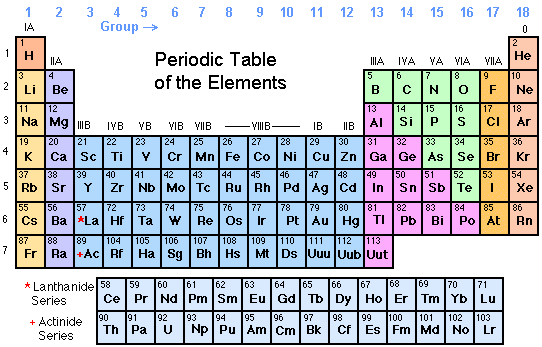
Some of the metalloids, such as silicon and germanium, are semi-conductors. Metalloids have properties of both metals and non-metals. Metalloids are the elements found between the boundary that distinguishes metals from non-metals. They have oxidation numbers of +3, ±4, and -3. All of these elements are solid, have a relatively high density, and are opaque. These elements, unlike the transition elements, do not exhibit variable oxidation states, and their valence electrons are only present in their outer shell. While these elements are ductile and malleable, they are not the same as the transition elements. The "other metals" elements are located in groups 13, 14, and 15. This is why they often exhibit several common oxidation states. Their valence electrons are present in more than one shell. The 38 elements in groups 3 through 12 of the periodic table are called "transition metals." As with all metals, the transition elements are both ductile and malleable, and conduct electricity and heat. All alkaline earth elements have an oxidation number of +2, making them very reactive. The alkaline earth elements are metallic elements found in the second group of the periodic table. The alkali metals are softer than most other metals. As with all metals, the alkali metals are malleable, ductile, and are good conductors of heat and electricity. Therefore, they are ready to lose that one electron in ionic bonding with other elements. These metals have only one electron in their outer shell. The alkali metals, found in group 1 of the periodic table, are highly reactive metals that do not occur freely in nature. For our purposes we will define the following ten families: The Periodic table can also be divided into several families of elements each having similar properties. Elements of each group had similar properties. The modern one states that the properties vary with atomic number, not weight.Įlements in Mendeleev's table were arranged in rows called periods. 
His periodic law states that the chemical and physical properties of the elements vary in a periodic way with their atomic weights. He observed that many elements had similar properties, and that they occur periodically. Mendeleev created the first periodic table based on atomic weight. 
They both created similar periodic tables only a few months apart in 1869. The modern periodic table, based on atomic number and electron configuration, was created primarily by a Russian chemist, Dmitri Ivanovich Mendeleev, and a German physicist, Julius Lothar Meyer, both working independently. The valence electrons are held closer towards the nucleus of the atom.Chemistry and Periodic Table Applications This means that the nucleus attracts the electrons more strongly, pulling the atom's shell closer to the nucleus. The effect of increasing proton number is greater than that of the increasing electron number therefore, there is a greater nuclear attraction. However, at the same time, protons are being added to the nucleus, making it more positively charged. 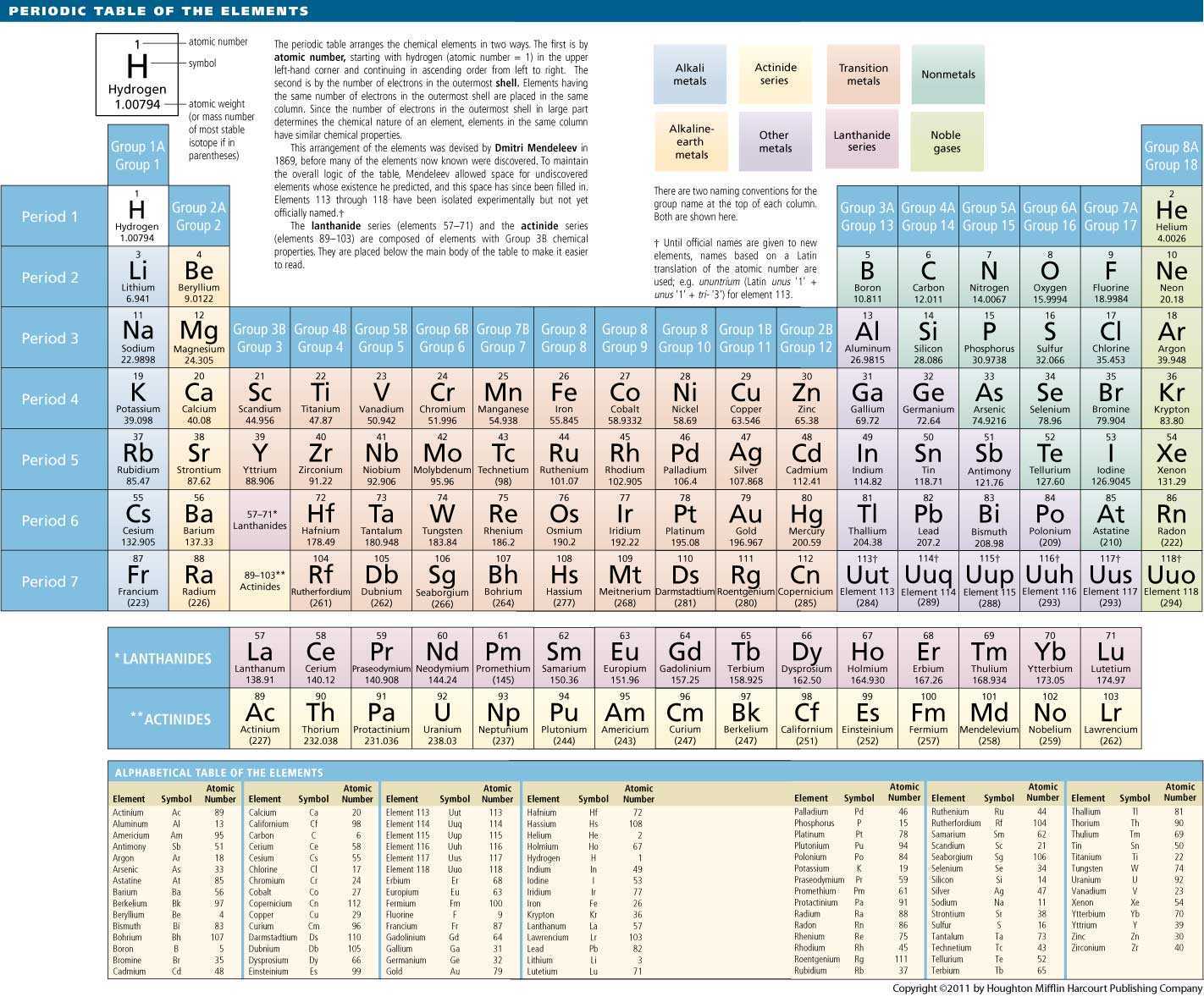
This is because, within a period or family of elements, all electrons are added to the same shell. Atomic radius patterns are observed throughout the periodic table.Ītomic size gradually decreases from left to right across a period of elements. The covalent radii of these molecules are often referred to as atomic radii. Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). This is caused by the increase in atomic radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
